Abstract
Research Article
Assessing the Neurocognitive function effects of ketamine in Cardiac Surgical patients
Demet Dogan Erol*
Published: 23 October, 2018 | Volume 2 - Issue 1 | Pages: 018-022
Background: Despite remarkable progress in surgical, cardiopulmonary bypass (CPB) and anesthetic tecniques, neurocognitive damage still remains an important cause of postoperative morbidity in cardiac surgery. The aetiology of neurocognitive damage is likely to be multifocal; including macro and microemboli, cerebral hypoperfusion, inflammation and nonpulsatile flow. N-methyl-D-asparticAcid (NMDA) receptors play an important role during neurocognitive damage. Ketamine is a non-competitive antagonist to the phencyclidine site of NMDA receptor for glutamate and directly suppresses proinflammatory cytokine production. The aim of the present study was to evaluate whether ketamine has neuroprotective effects during open-heart surgery through the use of neurocognitive tests.
Methods: We considered all patients aged between 58-76 years who were referred to a single cardiothoracic surgical team for elective, primary coronary revascularization. Patients were excluded from the study for the following reasons: a history of neurological, psychiatric, gastrointestinal, hepatic, renal, hematologic and clotting systems disorder and repeat procedures. Undergoing CPB were randomized 2 groups: Group1 (ketamine)(n=25) or Group2 (propofol)(n= Patients 25) In the propofol group, anesthesia was induced with 3mg/kg propofol, 1µg/kg remifentanyl, 0.1mg/kg vecuronium. Remifentanyl 0.5-1μg/kg/min was infused intravenously throughout the whole procedure. In the ketamine group, anesthesia was induced with 1-2mg/kg propofol, 1-2mg ketamin, 0.1mg/kg vecuronium. Ketamin 1mg/kg/hour was infused intravenously. Pressors, inotropic agents and antiarrhythmics were used as needed. The Mini-Mental State Examination(MMSE) was administered the day before surgery and three days later. The change in scores for MMSE was calculated for each patient and all the group. The results were compared statistically with paired simple t-test.
Results: The mean age, CBP duration, lowest temperature was not statistically significant (Table1). Peroperative and postoperative blood pressures and pulse rates showed differences between groups. There were no preoperative differences between the groups on any of the mean MMSE score (Table2). The ECG monitoring revealed that most patients remained in sinus rhythm, with no difference between groups.
Conclusions: We could not demonstrate that intraoperatively administered ketamine resulted in greater neuroprotective effects compared with propofol. Ketamine in combination with propofol during cardiac surgery is associated with a stable hemodynamic profile. Propofol may reduce the delivery of microemboli to the cerebral circulation by decreasing the cerebral blood flow. Propofol has a direct neuroprotective effect in vitro, although Roach et al. could not demonstrate a protective effect of propofol during open-heart surgery. Propofol enhances the antiinflammatory response to surgery by several mechanisms. This might have masked a neuroprotective effect of ketamine because propofol was administered in both groups in our study.
Read Full Article HTML DOI: 10.29328/journal.ijcar.1001007 Cite this Article Read Full Article PDF
Keywords:
Cardiac surgical procedures; Cardiopulmonary bypass; General anesthesia; Ketamine; Propofol; Neuroprotective agents
References
- Lombard FW, Mathew JP. Neurocognitive dysfunction following cardiac surgery. Sem Cardiothorac Vasc Anesth. 2010; 14: 102-110. Ref.: https://goo.gl/4R41sv
- Arrowsmith J, Grocott H, Reves J, Newman MF. Central nervous system complications of cardiac surgery. Br J Anaesth. 2000; 84: 378–393. Ref.: https://goo.gl/4WJr1t
- Pugsley W, Klinger L, Paschalis C, Treasure T, Harrison M, et al. The impact of microemboli in cardiopulmonary bypass on neuropsychological functioning. Stroke. 1994; 25: 1393–1399. Ref.: https://goo.gl/Ya8K6Z
- Smith QR. Transport of glutamate and other amino acids at the blood-brain barrier. J Nutr. 2000; 130: 1016-1022. Ref.: https://goo.gl/eCC29Q
- Kawasaki T, Ogata M, Kawasaki C, Ogata J, Inoue Y, et al. Ketamine suppresses proinflammatory cytokine production in human whole blood in vitro. Anesth Analg. 1999; 89: 665–669. Ref.: https://goo.gl/3tmdxB
- Nagels W, Demeyere R, Hemelrijck JV, Vandenbussche E, Gijbels K, et al. Evaluation of the Neuroprotective Effects of S(+)-Ketamine During Open-Heart Surgery. Anesth Analg. 2004; 98: 1595-1603. Ref.: https://goo.gl/cqVBqe
- Hindman BJ, Todd MM. Improving Neurologic Outcome after Cardiac Surgery. Anesthesiology. 1999; 90: 1243-1247. Ref.: https://goo.gl/zVU3wU
- Laffey JG, Boylan JF, Cheng DC. The systemic inflammatory response to cardiac surgery. Anesthesiology. 2002; 97: 215–252. Ref.: https://goo.gl/qcTDq2
- Smith P, Treasure T, Newman SP, Joseph P, Ell PJ, et al. Cerebral consequences of cardiopulmonary bypass. Lancet. 1986; 1: 823–825. Ref.: https://goo.gl/RZwu4J
- Gladstone DJ, Black SE, Hakim AM; Heart and Stroke Foundation of Ontario Centre of Excellence in Stroke Recovery. Toward Wisdom from Failure: Lessons from Neuroprotective Stroke Trials and New Therapeutic Directions. Stroke. 2002; 33: 2123 - 2136. Ref.: https://goo.gl/GRZ34D
- Newman MF, Murkin JM, Roach G, Croughwell ND, White WD, et al. Cerebral physiologic effects of burst suppression doses of propofol during nonpulsatile cardiopulmonary bypass: CNS Subgroup of McSPI. Anesth Analg. 1995; 81: 452–457. Ref.: https://goo.gl/yLm8bV
- Hollrigel G, Toth K, Soltesz I. Neuroprotection by propofol in acute mechanical injury: role of GABAergic inhibition. J Neurophysiol. 1996; 76: 2412–2422. Ref.: https://goo.gl/h8oUmn
- Bhudia SK, Cosgrove DM, Naugle RI, Rajeswaran J, Buu-Khanh Lam, et al. Magnesium as a neuroprotectant in cardiac surgery: A randomized clinical trial. J Thorac Cardiovasc Surg. 2006; 131: 853-861. Ref.: https://goo.gl/g737xX
- Butterworth J, Hammon JW. Lidocaine for Neuroprotection: More Evidence of Efficacy. Anesth Analg. 2002; 95: 1131-1133. Ref.: https://goo.gl/Y2Nw8F
- Arrowsmith JE, Harrison MJG, Newman SP, Stygall J, Timberlake N, et al. Neuroprotection of the Brain During Cardiopulmonary Bypass A Randomized Trial of Remacemide During Coronary Artery Bypass in 171 Patients. Stroke. 1998; 29: 2357–2362. Ref.: https://goo.gl/4Dy7D4
- Ozyurt E, Graham DI, Woodruff GN, Culloch J. Protective effect of the glutamate antagonist MK801 in focal cerebral ischaemia in the cat. J Cereb Blood Flow Metab. 1988; 8: 138–143. Ref.: https://goo.gl/TSzGxt
- Bruno V, Copani A, Knöpfel T, Kuhn R, Casabona G, et al. Activation of metabotropic glutamate receptors coupled to inositol phospholipid hydrolysis amplifies NMDA-induced neuronal degeneration in cultured cortical cells. Neuropharmacology. 1995; 34: 1089-1098. Ref.: https://goo.gl/ZFdjQ5
- Burker EJ, Blumenthal JA,Feldman M, Thyrum E, Mahanna E, et al. The Mini Mental State Exam as a predictor of neuropsychological functioning after cardiac surgery. Int J Psychiatry Med 1995; 25: 263-276. Ref.: https://goo.gl/Ciet9a
- Lewis MS, Maruff PT, Silbert BS. Examination of the Use of Cognitive Domains in Postoperative Cognitive Dysfunction after Coronary Artery Bypass Graft. Surgery. Ann Thorac Surg 2005; 80: 910 - 916. Ref.: https://goo.gl/kUPVNq
- Weissrock S, Levy F, Balabaud V, Thiranos JC, Dupeyron JP, et al. Interest of the Mini Mental State Examination to detect cognitive defects after cardiac surgery. Ann Fr Anesth Reanim. 2005; 24: 1255-1261. Ref.: https://goo.gl/BmesV8
Figures:
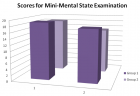
Figure 1
Similar Articles
-
Evolution of anaesthesia in transapical aortic valve implantation Running head: Anaesthesia and transcatheter valveMarzia Cottini*, Lappa A,Donfrancesco S,Francesco Musumeci. Evolution of anaesthesia in transapical aortic valve implantation Running head: Anaesthesia and transcatheter valve . . 2017 doi: 10.29328/journal.hacr.1001003; 1: 020-023
-
Assessing the Neurocognitive function effects of ketamine in Cardiac Surgical patientsDemet Dogan Erol*. Assessing the Neurocognitive function effects of ketamine in Cardiac Surgical patients . . 2018 doi: 10.29328/journal.ijcar.1001007; 2: 018-022
-
Effects of intraoperative epidural anesthesia during hepatectomy on intraoperative and post-operative patient outcomesChristopher Mallard,Brad Withers,Brooke Bauer,Paul A Sloan,Sean Dineen,Annette Rebel*. Effects of intraoperative epidural anesthesia during hepatectomy on intraoperative and post-operative patient outcomes. . 2018 doi: 10.29328/journal.ijcar.1001008; 2: 023-030
-
The choice of optimal modern muscle relaxants (rocuronium bromide, atracurium besilate and cisatracurius besilate) in one-day surgery in childrenNasibova EM*. The choice of optimal modern muscle relaxants (rocuronium bromide, atracurium besilate and cisatracurius besilate) in one-day surgery in children. . 2020 doi: 10.29328/journal.ijcar.1001014; 4: 004-012
-
An Instance of Green-tinted Urine Related to the use of PropofolBindhya Maharjan*, Jeevan Singh, Shibesh Chandra Mishra, Saubhagya Neupane. An Instance of Green-tinted Urine Related to the use of Propofol. . 2024 doi: 10.29328/journal.ijcar.1001024; 8: 001-004
-
Anesthetic Management of a Patient with Left Ventricular Thrombus Posted for Emergency LaparotomyArpita Das*. Anesthetic Management of a Patient with Left Ventricular Thrombus Posted for Emergency Laparotomy. . 2025 doi: 10.29328/journal.ijcar.1001028; 9: 013-016
Recently Viewed
-
Do Fishes Hallucinate Human Folks?Dinesh R*,Sherry Abraham,Kathiresan K,Susitharan V,Jeyapavithran C,Paul Nathaniel T,Siva Ganesh P. Do Fishes Hallucinate Human Folks?. Arch Food Nutr Sci. 2017: doi: 10.29328/journal.afns.1001003; 1: 020-023
-
Assessment of Redox Patterns at the Transcriptional and Systemic Levels in Newly Diagnosed Acute LeukemiaAna Carolina Agüero Aguilera, María Eugenia Mónaco, Sandra Lazarte, Emilse Ledesma Achem, Natalia Sofía Álvarez Asensio, Magdalena María Terán, Blanca Alicia Issé, Marcela Medina, Cecilia Haro*. Assessment of Redox Patterns at the Transcriptional and Systemic Levels in Newly Diagnosed Acute Leukemia. J Hematol Clin Res. 2024: doi: 10.29328/journal.jhcr.1001029; 8: 017-023
-
Assessment of Indigenous Knowledge on Using of Traditional Medicinal Plants to Cure Human Diseases in South Omo Zone Baka Dawla Ari District, Kure and Bitsmal South EthiopiaGizaw Bejigo*. Assessment of Indigenous Knowledge on Using of Traditional Medicinal Plants to Cure Human Diseases in South Omo Zone Baka Dawla Ari District, Kure and Bitsmal South Ethiopia. J Plant Sci Phytopathol. 2024: doi: 10.29328/journal.jpsp.1001132; 8: 048-054
-
Nanoencapsulated Extracts from Leaves of Bauhinia forficata Link: In vitro Antioxidant, Toxicogenetic, and Hypoglycemic Activity Effects in Streptozotocin-induced Diabetic MiceBárbara Verônica Cardoso de Souza, Alessandra Braga Ribeiro*, Rita de Cássia Meneses Oliveira, Julianne Viana Freire Portela, Ana Amélia de Carvalho Melo Cavalcante, Esmeralda Maria Lustosa Barros, Luís Felipe Lima Matos, Tarsia Giabardo Alves, Maria. Nanoencapsulated Extracts from Leaves of Bauhinia forficata Link: In vitro Antioxidant, Toxicogenetic, and Hypoglycemic Activity Effects in Streptozotocin-induced Diabetic Mice. Arch Pharm Pharma Sci. 2024: doi: 10.29328/journal.apps.1001063; 8: 100-115
-
GELS as Pharmaceutical Form in Hospital Galenic Practice: Chemico-physical and Pharmaceutical AspectsLuisetto M*,Edbey Kaled,Mashori GR,Ferraiuolo A,Fiazza C,Cabianca L,Latyschev OY. GELS as Pharmaceutical Form in Hospital Galenic Practice: Chemico-physical and Pharmaceutical Aspects. Arch Surg Clin Res. 2025: doi: 10.29328/journal.ascr.1001084; 9: 001-007
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."